
IATF 16949 Clause 10.2.3 — Problem Solving — holds the top position in the IAOB’s major nonconformity rankings at 10.31% of all major NCs across 19 IATF certification bodies, running nearly double the frequency of the second-ranked clause. Organisations aren’t failing because they lack 8D templates or problem-solving training. They’re failing because their root cause analysis identifies symptoms and presents them as causes — and the standard itself provides no evidential threshold to distinguish between the two.
What IATF 16949 Clause 10.2.3 Actually Requires
IATF 16949:2016 Clause 10.2.3 mandates a documented problem-solving process covering six sub-requirements. Three generate the most audit exposure.
Sub-clause (c) requires root cause analysis with documented methodology, analysis, and results. The critical word is “results” — the submission must contain evidence of the causal link between the identified root cause and the nonconformity, not a narrative assertion that one caused the other. An 8D that states “the root cause was inadequate operator training” at D4 without production records, control data, or process evidence linking that training gap to the specific defect mechanism has documented methodology and analysis but not results.
Sub-clause (d) requires implementation of systemic corrective actions, including consideration of the impact on similar processes and products — meaning a corrective action that addresses a single production line without a documented assessment of whether the same root cause exists across other lines, product families, or facilities doesn’t satisfy what the standard demands.
Sub-clause (f) requires updating appropriate documented information — the process FMEA and control plan — where necessary, and this sub-clause alone accounts for 40.83% of all Clause 10.2.3 nonconformances in the IAOB’s NC-CARA dataset because the correction happened on the floor but the process control documentation was never revised to reflect it.
Clause reference reflects mapped standard requirement. Verify against current edition before audit use.
Where Organisations Fail on Root Cause Analysis
The IAOB’s AI-assisted analysis of its 19-CB NC-CARA database pinpointed the primary objective evidence driver for Clause 10.2.3 NCs: investigations that terminate at symptom-level identification rather than reaching systemic causation.
Here’s what a rejected submission looks like in practice.
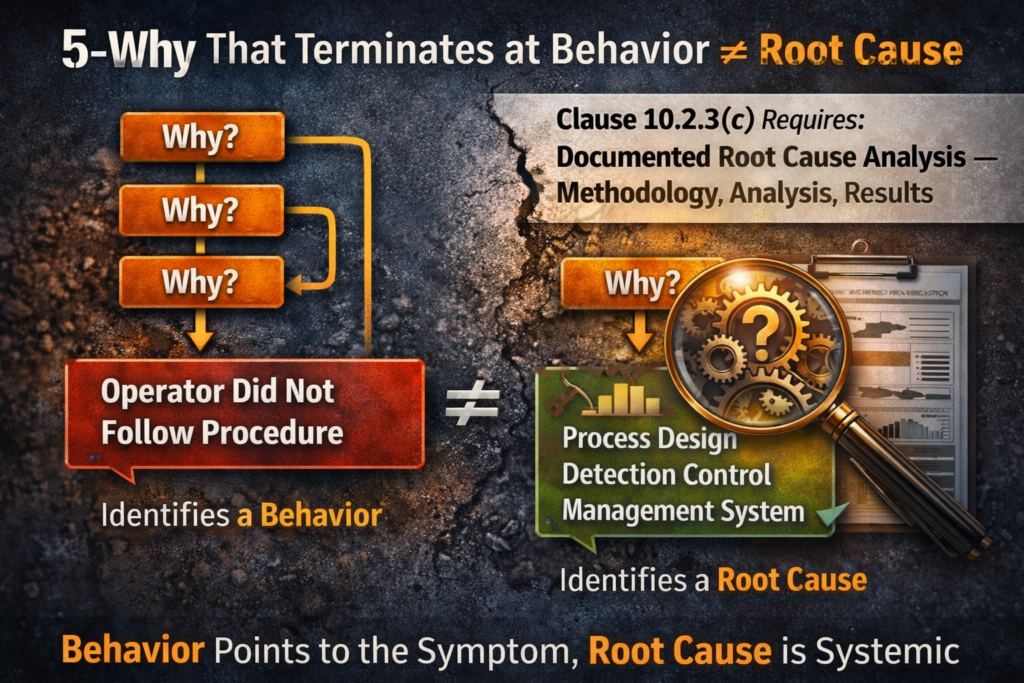
The 5-Why chain reaches “operator did not follow procedure” at Why 3 and stops. Nobody asks what detection control failed to catch the deviation, what process design permitted the deviation to produce a defect, or what management oversight mechanism should have caught the drift before it reached the customer. The auditor writes it up: root cause is a symptom, systemic corrective action not demonstrated.
The NC-CARA data reveals something worse. Organisations submitting corrective actions to the database are self-reporting root causes that confirm the pattern — inadequate training, procedural gaps, insufficient management oversight — and these self-reported causes are themselves symptom-level, which means the organisations investigating their own problem-solving failures are reproducing the exact analytical error that generated the nonconformity in the first place.
A second failure mode compounds the first. Clause 10.2.1(d) requires the organisation to review the effectiveness of corrective action taken, and this sub-clause accounts for 60.45% of all Clause 10.2.1 nonconformances. Corrective actions get closed at implementation — “we retrained the operators” — with no measurement period defined and no data collection plan. Six months later at surveillance, the auditor requests effectiveness evidence. None.
What an Audit-Defensible Submission Looks Like
An audit-defensible Clause 10.2.3 submission contains four elements that most rejected submissions lack.
First: a root cause statement identifying a mechanism, not a behaviour. “The fixture alignment tolerance was set at ±0.5mm but process capability data shows Cpk of 0.87 against the customer specification” is a mechanism. “The operator did not check the fixture” is a behaviour — it describes who was present when the defect occurred, not why the defect was possible. Auditors care about the mechanism.
Second: evidence validating the root cause — production records, SPC data, inspection logs, or shift records confirming the identified mechanism was active during the period when nonconformities were detected, because a hypothesis without supporting evidence is an assertion, and Clause 10.2.3(c) requires results.
Third: a documented systemic impact review covering other processes, product families, and other facilities where the same mechanism could exist, with the scope of the review, the method used, and the result recorded even if the result is “no impact identified in Lines 2 through 4.”
Fourth: revised pFMEA and control plan referencing the corrective action before the 8D is closed, with the failure mode appearing in the FMEA carrying updated severity, occurrence, and detection ratings, and the control plan reflecting whatever monitoring, inspection, or error-proofing was added. Without this step, the correction exists in the quality record but not in the process control architecture — and that’s the 40.83% gap.
Practical Steps to Fix Your 8D Process
- Define the causal standard before opening D4. The cross-functional team agrees on one rule: an acceptable root cause identifies a process, control, or system failure — not a human behaviour. Any root cause that a person could repeat even after retraining hasn’t reached the systemic layer.
- Apply the systemic layer test at Why 3. Every time the analysis chain reaches “operator error,” “lack of training,” or “procedure not followed,” add one more Why — what process design, detection mechanism, or management system permitted this behaviour to produce a defect without being caught, and why did the existing controls fail to flag it before the product shipped?
- Validate before closing D4. Collect objective evidence — production records, monitoring data, shift logs — confirming the identified mechanism was active during the nonconformity period. If the evidence doesn’t support the hypothesis, the root cause is wrong. Find another.
- Execute the systemic impact review before D5. Document which processes, products, and facilities were assessed, the method used, and the result. Clause 10.2.3(d) requires it. The auditor will ask.
- Update pFMEA and control plan before claiming closure. Assign this at D5 with a named owner and deadline — don’t wait until after verification, because the 40.83% sub-clause failure rate exists precisely because organisations treat documentation updates as something they’ll get to later.
- Build the effectiveness verification plan at D6 by specifying the metric, the monitoring period, and the go/no-go criterion that will determine whether the corrective action actually eliminated the root cause or merely suppressed the symptom. At surveillance, this plan and its results are the Clause 10.2.1(d) evidence package.

The Coverage Gap Underneath Clause 10.2.3
There is a structural reason IATF 16949 Clause 10.2.3 persists as the #1 nonconformity despite universal awareness of 8D, 5-Why, and Ishikawa methods.
The standard mandates problem-solving methodology and root cause analysis but defines no minimum adequacy criteria — no sub-clause specifies what causal depth qualifies, no Sanctioned Interpretation addresses what separates a valid root cause from a symptom statement, and no AIAG guidance document draws the evidential boundary.
The practical result: the CB auditor’s professional judgment is the operative standard for root cause adequacy, and across 19 certification bodies, that judgment isn’t uniform.
An organisation can’t audit-proof a submission against a requirement that has no defined evidential threshold. What it can do is make the submission self-evidently causal — build the evidence package so the distinction between mechanism and symptom is obvious on its face.
That’s the real quality management challenge here. Not completing the 8D form. Not picking the right problem-solving tool. Producing a root cause analysis that would survive scrutiny from any auditor in the 19-CB landscape, because the evidence demonstrates the causal link rather than merely asserting it.
Clause reference reflects mapped standard requirement. Verify against current edition before audit use.
Frequently Asked Questions
Q: What is the most common IATF 16949 nonconformity? A: Clause 10.2.3 — Problem Solving — holds the top position at 10.31% of all major nonconformities across the 19 IATF certification bodies, nearly double the frequency of the second-ranked clause.
Q: Why do root cause analyses fail IATF audits? A: Most failures terminate at symptom-level identification — citing human behaviours like “operator did not follow procedure” — rather than reaching the systemic process, control, or design mechanism that permitted the defect to occur and escape detection.
Q: What must be updated before closing an 8D under Clause 10.2.3? A: The process FMEA and control plan must be revised to reflect the corrective action, with updated severity, occurrence, and detection ratings in the FMEA and any added monitoring or error-proofing documented in the control plan.
Q: What is the systemic impact review required by Clause 10.2.3(d)? A: A documented assessment of whether the identified root cause mechanism exists across other production lines, product families, or facilities, recording the scope, method, and result — even if no impact is found.
About AEC International
AEC International provides ISO certification, training, and consultancy services at the intersection of automotive quality management and operational excellence. We support organisations across the automotive supply chain in achieving and maintaining IATF 16949 certification — from gap analysis and implementation through audit preparation and continual improvement.
Learn more: www.aec.llc
