Tag
Legal Compliance

ISO 13485 Clause 7.3 Exclusion: Why Your Design Controls Justification Fails Under the FDA QMSR
Clause 7.3 exclusion under FDA QMSR is no longer a settled historical decision. Contract manufacturers and component suppliers must update ISO 13485 quality manual justifications, reassess design activity boundaries, and prepare current audit evidence.
Read article →

ISO 42001 Statement of Applicability and Impact Assessment: Two Errors That Survive Audit Preparation
Organisations building their first ISO/IEC 42001:2023 AI Management System are importing assumptions from ISO 27001 that do not transfer. Two structural errors reach Stage 2 audit preparation uncorrected:…
Read article →

ISO 14001 Clause 6.1.2: Why Your Aspect Register’s Life Cycle Perspective Probably Stops Too Soon
Most ISO 14001 aspect registers cover site-boundary operations but omit upstream and downstream life cycle stages without documented rationale. Clause 6.1.2 requires documented consideration of every stage — exclusion needs written evidence, not silence.
Read article →
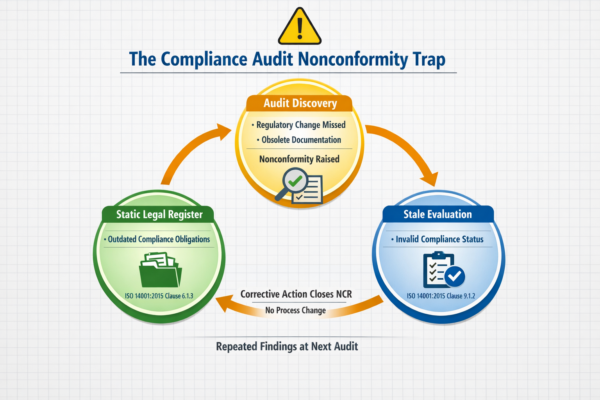
Why Your ISO 14001 Legal Register Fails Surveillance Audits
Static ISO 14001 legal registers generate linked nonconformities across Clauses 6.1.3 and 9.1.2. This article diagnoses why registers go stale, what auditors actually probe during surveillance, and how to rebuild before the next audit cycle.
Read article →

ISO 13485 Design Controls Under FDA QMSR: Why Relabeled DHF Records Fail at Clause 7.3
Relabeled DHF headers do not satisfy ISO 13485 design controls under FDA QMSR. This analysis identifies the three Clause 7.3 gaps — at 7.3.3, 7.3.6, and 7.3.7 — where legacy 820.30 records break, and provides a phased implementation sequence to close them.
Read article →
