Tag
Risk Management

ISO 13485 Clause 7.3 Exclusion: Why Your Design Controls Justification Fails Under the FDA QMSR
Clause 7.3 exclusion under FDA QMSR is no longer a settled historical decision. Contract manufacturers and component suppliers must update ISO 13485 quality manual justifications, reassess design activity boundaries, and prepare current audit evidence.
Read article →

ISO 37001 “Reasonable and Proportionate”: Why Uniform Controls Fail the Standard’s Own Test
Quick Answer: ISO 37001:2025 requires every anti-bribery control to be calibrated to documented bribery risk — not applied uniformly. “Reasonable and proportionate” is a design instruction: risk assessment…
Read article →

ISO 42001 Statement of Applicability and Impact Assessment: Two Errors That Survive Audit Preparation
Organisations building their first ISO/IEC 42001:2023 AI Management System are importing assumptions from ISO 27001 that do not transfer. Two structural errors reach Stage 2 audit preparation uncorrected:…
Read article →

ISO 22301 Clause 8.4: Why Plan-Centric BCMS Implementations Fail Under Exercise Testing
ISO 22301 Clause 8.4 requires business continuity plans built on BIA outputs and selected strategies. Organisations that start with the plan bypass the Clause 8 dependency chain and produce documents that fail under exercise testing.
Read article →

ISO 27001:2022 Clause 6.1.2: Why Your Carried-Forward Risk Assessment Methodology Is an Audit Liability
**Excerpt:** ISO 27001:2022 removed asset/threat/vulnerability as a normative prerequisite for risk identification. Organisations that carried their methodology forward without documenting the choice under Clause 6.1.2 now face a Major nonconformity risk at their next audit. (42 words)
Read article →

ISO 45001 Clause 4.2: Why Your Interested Parties Register Isn’t What the Standard Requires
Most ISO 45001 Clause 4.2 registers list workers and generic needs but never feed into objectives. This article traces the structural dependency from Clause 4.2 through worker consultation under 5.4 to objective-setting under 6.2 — and shows how to close the gap before auditors do.
Read article →

ISO 14001 Clause 6.1.2: Why Your Aspect Register’s Life Cycle Perspective Probably Stops Too Soon
Most ISO 14001 aspect registers cover site-boundary operations but omit upstream and downstream life cycle stages without documented rationale. Clause 6.1.2 requires documented consideration of every stage — exclusion needs written evidence, not silence.
Read article →

Your Risk Register Doesn’t Satisfy ISO 9001 Clause 6.1 — Here’s What Does
Most ISO 9001 risk registers list risks without changing anything downstream. Clause 6.1 conformance requires traceable integration into process controls and quality objectives — a gap ISO DIS 9001:2025 will make structurally visible.
Read article →

Your Internal Audit Programme Isn’t Risk-Based — And ISO 9001’s Revision Will Prove It
Most ISO 9001 audit programmes run fixed-rotation schedules with no risk-based frequency rationale. ISO DIS 9001:2025 adds defined per-audit objectives — exposing the structural gap. Here's how to rebuild before transition.
Read article →
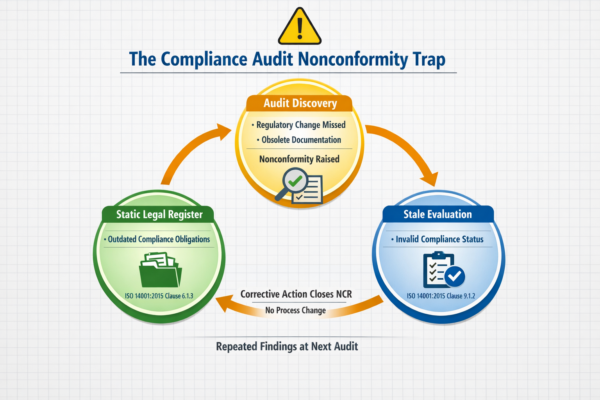
Why Your ISO 14001 Legal Register Fails Surveillance Audits
Static ISO 14001 legal registers generate linked nonconformities across Clauses 6.1.3 and 9.1.2. This article diagnoses why registers go stale, what auditors actually probe during surveillance, and how to rebuild before the next audit cycle.
Read article →

ISO 50001 Energy Baseline Drift: Why Your EnPIs Won’t Survive CSRD Assurance
ISO 50001 energy baseline drift produces EnPIs that look like improvement but won't survive CSRD limited assurance. Learn why Clause 6.5 gaps create cross-framework reporting risk and how to build a baseline review trigger protocol.
Read article →
